The objective of this study was to evaluate the performance and mechanical integrity of the
OSSIOfiber® Compression Screw in a full load-bearing osteotomy fixation in an ovine model through 6 months.
Key Takeaways:
- At 2 weeks post-operative, all subjects returned to normal gait and function with X-rays demonstrating stable fixation and good anatomical alignment
- At 3 months post-operative, all animals demonstrated bony fusion with 100% bone bridging across osteotomy sites, well maintained reduction of the osteotomy and no evidence of implant migration
- At 6 months post-operative, all animals showed optimal biocompatibility, maintained structural integrity of the implants and excellent bio-integration with early mesenchymal cellular investment into the implant
Histopathology results at 6 months indicated absence of callus formation, representing healing by first intention, was indicative of a very stable construct, with no signs of implant loosening or migration.
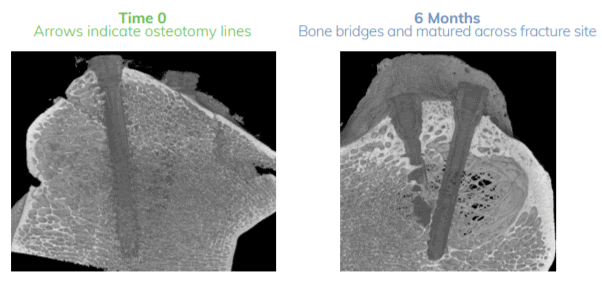
Figure: Micro CT Imaging – Time 0 compared to 6-months post-operative demonstrate
accurate reduction and excellent bone healing and fusion.
Discover the comprehensive reviews, X-rays, and micro-CT scans throughout the 6 month study.
